ETC
-
Paxerontine SR Tab. 12.5mg
- Ingredient
Paroxetine Hydrochloride
- Content
12.5mg
- Indication
1) Treatment of major depressive disorder
2) Treatment of panic disorder, with or without agoraphobia
3) Treatment of social anxiety disorder, also known as social phobia
4) Treatment of premenstrual dysphoric disorder (PMDD)
-
Paxerontine SR Tab. 25mg
- Ingredient
Paroxetine Hydrochloride
- Content
25mg
- Indication
1) Treatment of major depressive disorder
2) Treatment of panic disorder, with or without agoraphobia.
3) Treatment of social anxiety disorder, also known as social phobia.
4) Treatment of premenstrual dysphoric disorder (PMDD).
-
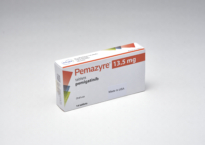
Pemzayre® tablet 13.5mg
- Ingredient
1 Tab(240.0mg) contains, pemigatinib (In-house spec.) 13.5mg
- Content
White to off-white round tablet, one side debossed with “I”, the other side with “13.5”.
- Indication
The treatment of adults with locally advanced or metastatic cholangiocarcinoma with a fibroblast growth factor receptor 2 (FGFR2) fusion or rearrangement that have progressed after one or more prior line of systemic therapy.
-
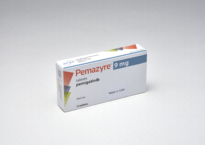
Pemzayre® tablet 9mg
- Ingredient
1 Tab(160.0mg) contains, pemigatinib (In-house spec.) 9mg
- Content
White to off-white oval tablet, one side debossed with “I”, the other side with “9”.
- Indication
The treatment of adults with locally advanced or metastatic cholangiocarcinoma with a fibroblast growth factor receptor 2 (FGFR2) fusion or rearrangement that have progressed after one or more prior line of systemic therapy.
-
Pemzyre® tablet 4.5mg
- Ingredient
1 Tab(80.0mg) contains, pemigatinib (In-house spec.) 4.5mg
- Content
White to off-white round tablet, one side debossed with “I”, the other side with “4.5”.
- Indication
The treatment of adults with locally advanced or metastatic cholangiocarcinoma with a fibroblast growth factor receptor 2 (FGFR2) fusion or rearrangement that have progressed after one or more prior line of systemic therapy.